| CHIRAL
MESOGENS |
|
Chiral
liquid crystal molecules have the potential to form chiral mesophases.
In fact, it was quite a surprise to discover that DNA (which is highly
chiral) could form achiral line hexatic phases.(1,2) Normally,
chiral molecules lead to chiral phases, where the director twists as
it progresses through the phase.
|
| CHIRAL
NEMATIC PHASE (CHOLESTERIC) |
 |
If
a nematic liquid crystal is inherently chiral, then adjacent mesogens
will have a preferential twist with respect to one another, which
leads to the larger-scale twisting of the internal order. The
chiral nematic phase is also called the cholesteric phase
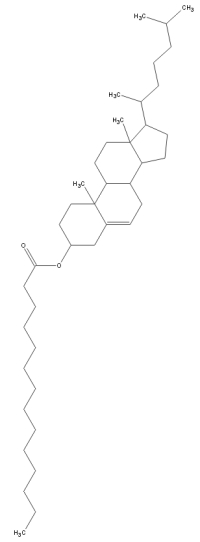
(since the phase was first discovered in cholesterols). The chemical
structure is of cholesteryl myristate is shown.
The
structure of the twisting nematic layers is shown below. The different
nematic layers have been coloured differently so as to differentiate
them. The twisting of the nematic director from layer to layer
is clear.

|
|
|
|
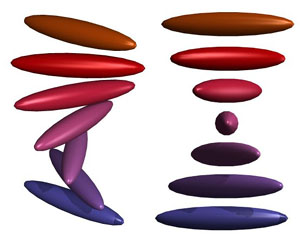
Beside
is a more idealized view of the nematic twisting of the cholesteric
phase (viewed both on-angle and dead-on). Clearly the cholesteric
phase is chiral, with a very specific handedness. This chiral
structure can be observed using optical techniques (for instance,
by viewing the phases through crossed polarizers).
|
|
| SMECTIC
C* |
|
The
chiral analogue of the Smectic C phase is denoted by a star (*).
In this phase, the tilt direction of the mesogens rotates as one
progresses through the layers. In the figure, the different layers
are coloured differently for convenience. The diagram to the far
right shows the twisting of the mesogens in a more idealized way,
and emphasizes the chiral twisting of the director.
|

|
|
|
|
|
| REFERENCES |
1.Podgornik,
R.; Strey, H.H.; Gawrisch, K.; Rau, D.C.; Rupprecht, A.; Parsegian,
V.A. Proc. Natl. Acad. Sci. USA 1996, 93, 4261.
2.Strey, H.H.; Wang, J.; Podgornik, R.; Rupprecht, A.; Yu, L.; Parsegian,
V.A.; Sirota, E.B. Phys. Rev. Lett. 2000, 84,
3105.
|
|
Return
to Barrett Research Group Homepage...
|



